A specialty chemical company publicizes a four-step protocol to test materials for resistance to disinfectants, drugs, and the other chemicals found in the hospital environment.
February 14, 2017
A specialty chemical company publicizes a four-step protocol to test materials for resistance to disinfectants, drugs, and the other chemicals found in the hospital environment.
In the medical device industry, the importance of choosing materials that can stand up to repeated exposure to harsh chemicals is becoming clear, as MD+DI has reported in the past. The use of strong disinfectants and rigorous sterilization procedures has intensified with healthcare reform and the focus on reducing hospital-acquired infections. Some plastics can crack, change color, become sticky, or even fail in these hospital environments, making chemical resistance a key factor in material selection.
"We really are seeing that a lot of our customers are having trouble with their device housings because of the amount of disinfection that's happening in the hospital," said Cynthia Lewis, market insight and strategy manager at Eastman Chemical Company. "Some of the bigger imaging equipment, for example, when they take that equipment out of service, that disrupts the revenue stream at the hospital," she added.
That's why Eastman, makers of materials for the medical device industry, has developed a 4-step testing protocol to examine the chemical resistance and impact strength of materials exposed to harsh disinfectants. Ellen Turner, market development manager in medical devices at Eastman, noted that while the device industry does already test materials, these tests don't always factor in the change in how disinfectants are used now.
In summary, these four steps are:
Use a jig with a strain level to accelerate stress cracking, such as 1.5%
Put plastic material flex bars on the jig
Apply a piece of cotton soaked in commonly used hospital chemicals to the flex bar and put the bars and jig in a closed plastic bag for 24 hours.
Take the samples off the jigs and observe any changes in the plastic. To complete the protocol, test the material bars for impact strength.
That last step, testing the plastic for impact strength retention, sets this protocol apart from other industry tests, Lewis explained. "The physical properties of materials used for a long time were chosen for impact strength, but the drugs and disinfectants are degrading that impact strength, which can't be seen visually . . . That hammer swinging down helps [testers] see that loss of impact strength after after chemical exposure," she said.
So what materials perform well? A chart from Eastman (below) shows that its Tritan material checks all the boxes. There are some materials that hold up fairly well throughout the first three steps of the protocol, Turner added. Polybutylene terephthalate (PBT) is one such material, she explained. "You're seeing now that polycarbonate is being blended with polyesters to improve its chemical resistance."
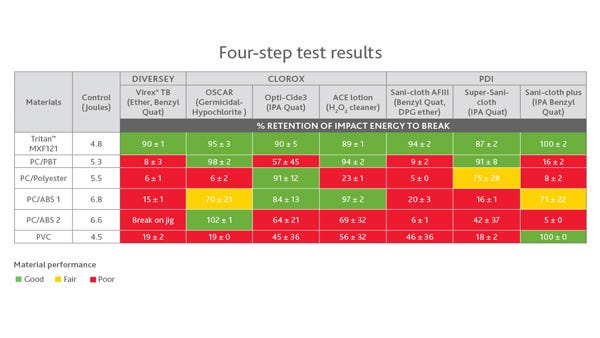
"We're excited because we have a new material, and we will have more new materials, based on better technology that resists contact with disinfectants and aggressive drugs--the environment that these things see in the hospital--not just a particular test we came up with because it's okay with our material," Turner said.
The Eastman protocol is intended as a resource for the industry as device makers prioritize chemical resistance in material selection. Turner said, "People are starving for a test method that makes more sense, that actually is applicable to the experience in the hospital, that's not so labor intensive, and that gives you a quick answer."
[Image courtesy of VITALWORKS/PIXABAY. Chart courtesy of EASTMAN CHEMICAL COMPANY]
About the Author(s)
You May Also Like


