ivWatch monitoring devices will now be compatible with Philips Intellivue Patient Monitors, enabling more healthcare facilities to use ivWatch devices to monitor peripheral IVs for infiltration and extravasation events.
July 5, 2018
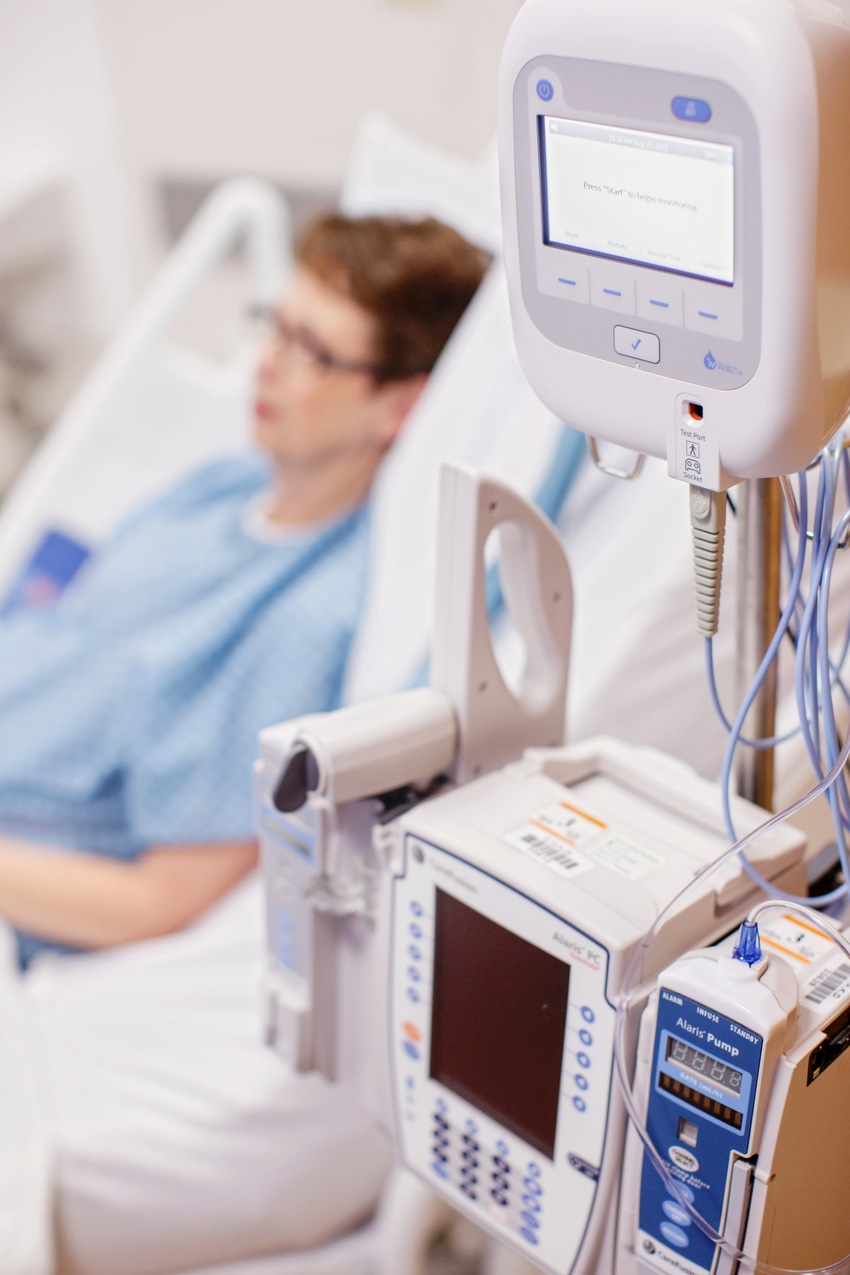
When it comes to peripheral intravenous (IV) therapy, doctors and nurses have come to accept that things might not go smoothly. Nearly 80% of hospital patients in the U.S. receive an IV — a procedure that has a startling 50% fail rate. ivWatch, the only provider of continuous non-invasive monitoring devices that can detect IV infiltrations and extravasations, hopes to curb patient harm from those numbers after announcing that their devices will now be compatible with Philips Intellivue Patient Monitors.
“For too long IV infiltration has been an accepted outcome of peripheral intravenous therapy,” says Gary Warren, president and CEO of ivWatch. “Similar to continuous monitors available for a patient’s heart and blood oxygen levels, ivWatch provides a first-of-its-kind, FDA-cleared, noninvasive medical device that continuously monitors peripheral IVs for infiltration and extravasation events. ivWatch has a standalone device (ivWatch Model 400) and comprehensive OEM solution that significantly improves the level of quality and patient safety through early detection of this common complication.”
The ivWatch Model 400 was designed to use light to monitor a patient’s subcutaneous tissue and can measure the changes in the optical properties. The device can essentially become a clinician’s partner by continuously monitoring for infiltrations, and sending an alert when the system detects an infiltration — enabling the clinician to inspect the IV site and intervene. With the company’s recent announcement, ivWatch technology can now issue alerts through Philips Intellivue Patient Monitors, a technology that has become ubiquitous throughout U.S. hospitals and clinics.
“ivWatch notifications will now appear in-room on Philips Patient Monitors and on remote nursing stations monitors, allowing clinicians to respond more quickly to possible IV infiltrations events,” Warren said. “IV assessment activity and infiltration/extravasation data can then be integrated into the electronic medical record (EMR), offering time-saving benefits, and a more holistic view of the patient.”
With a near 50% failure rate for IV therapy procedures, roughly 23% of those failures are due to infiltrations or extravasations, which occur when fluid or medication that is intended for the vein leaks into the surrounding tissue. Not only is this potentially harmful to the patient, it also leads to a medication dosing and drug delivery error that can prolong hospital stays and increase healthcare expenses. To date, most hospitals rely largely on educational programs to reduce severe safety events from IV infiltration and extravasation.
This growing problem is what led ivWatch to work with Philips to make the technology more available in hospitals and clinics around the U.S. Going forward, all of the ivWatch products will be compatible with the Philips IntelliBridge EC10 Interface Module, or IntelliBridge EC10 integral Interface Board with Open Interface Driver (ED101) and EC5 ID Module on MP40-90 and MX400-800 Philips patient monitors. All current ivWatch devices already on the market can also receive a software upgrade to support compatibility at no extra charge.
The company believes that their line of devices could be the difference between success and failure when it comes to peripheral IV therapy, with early detection being the crucial component to producing better patient outcomes.
“ivWatch exists to eliminate patient harm associated with infiltrations and extravasations, and we will not rest until patients around the world have access to this technology,” Warren said. “Early detection is key to keeping patients safe from harm. Our continuous monitoring solution aids in the early detection of peripheral IV infiltration, allowing for earlier intervention by the clinician. Having our technology support Philips IntelliVue Patient Monitors for in-room and remote notifications and EMR data capture will help ensure a practical implementation of ivWatch technology for our current and future customers.”
About the Author(s)
You May Also Like


