FDA cleared Abbott's FreeStyle Libre 3, which is expected to be the most accurate and smallest continuous glucose monitoring sensor on the market.
June 2, 2022
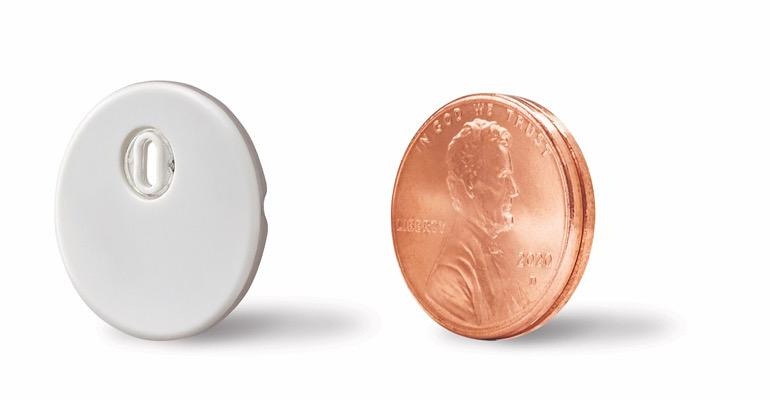
Just ahead of the American Diabetes Association's 82nd Scientific Sessions, Abbott nabbed FDA clearance for its FreeStyle Libre 3 system for people four years and older living with diabetes. FDA clearance comes nearly two years after the Libre 3 system received a CE mark for use in Europe.
"The FreeStyle Libre 3 system is a direct result of listening to our customers – and giving them the innovation and sensing technology they've been looking for," said Jared Watkin, senior vice president of Abbott's diabetes care business. "It's a game changer for the millions of people living with diabetes. They'll be able to manage their health minute-by-minute with the world's smallest and thinnest sensor and most accurate 14-day continuous glucose monitoring system."
The company notes that FreeStyle Libre 3 has a 7.9% overall mean absolute relative difference (MARD), making it the first continuous glucose monitoring (CGM) device to demonstrate a MARD below 8%. MARD is the standard way to meausre continuous glucose accuracy. Abbott also said the Libre 3 sensor is the smallest and thinnest on the market at the size of two stacked U.S. pennies. Other touted features include a one-piece applicator to apply te sensor, and a strong Bluetooth integration (rang up to 33 feet).
Abbott said it will price the FreeStyle Libre 3 system the same as previous versions.
"We continue to disrupt the notion that CGMs have to sacrifice quality or accuracy for affordability," said Watkin. "Access to breakthrough diabetes technologies should not be out of reach for the people who can benefit most from them. Innovation for access and affordability is a core pillar in Abbott's business and sustainability strategy."
The Abbott Park, IL-based company said the Libre 3 sensor will be available at participating pharmacies later this year. The system has been recognized multiple times as a top health tech innovation. It won the bronze award in the 2022 Medical Design Excellence Awards in the category of digital health products and mobile medical apps.
About the Author(s)
You May Also Like




