Cardiovascular
thumbnail
Cardiovascular
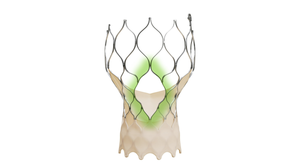
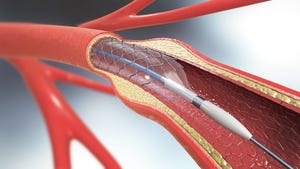
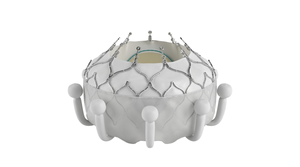
Is the SMART Trial a Game Changer for TAVR?How Will the SMART Trial Impact TAVR?
Analysts discuss the potential impact of a clinical trial that had Medtronic’s Evolut technology go head-to-head with Edward’s Sapien platform.
Sign up for the QMED & MD+DI Daily newsletter.










.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)




.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)


