April 18, 2016
| vs. | ||
|
|
|
|
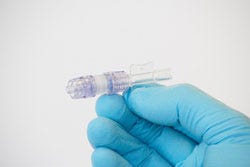
Describe your device and how it will benefit healthcare. | Clinical studies show that 7% of all I.V. lines are dislodged. In the United States alone, there are over 200 million I.V. lines placed annually. I.V. Dislodgment is estimated to cost the U.S. healthcare system nearly $1 billion a year. We are developing a new device to prevent catheter dislodgment called the SafeBreak IV. The current standard for preventing IV dislodgment is the use of tape or more expensive securement devices. While adhesives and securement are important, nothing can prevent dislodgment when a large force is applied to an IV line. The SafeBreak IV fits in-between the patient's catheter and the IV tubing, and creates a sealed liquid connection. Its design allows moderate pull force but separates before the adhesive fails, keeping harmful trauma from being delivered to the patient's IV site. When the device separates, its patented technology prevents blood loss and triggers the IV pump's high pressure alarm to alert the nurse. Replacing a SafeBreak IV is painless and takes seconds, so the patient can quickly resume their medication therapy without having to experience the pain of a dislodged IV and the reinsertion of a new one. The nursing staff can spend their time working on value-added activities and the hospital saves money on supplies, labor, and patient satisfaction. |
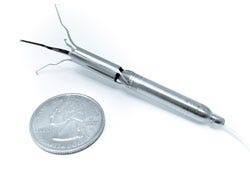
| Procyrion Inc. is working to give chronic heart failure patients a solution by developing the world's first catheter-deployed circulatory assist device intended for ambulatory use. The Aortix system, built around a powerful micromotor mounted on a set of expandable struts, is designed to rest the heart by reducing afterload and improve blood flow to vital organs. Unlike other circulatory assist devices, including ventricular assist devices (VADs), that involve invasive, high risk surgical procedures, Aortix is small enough--6 mm wide and less than 6.5 cm long--to allow for deployment without surgery and with minimal procedure risk. In a simple 20-minute procedure, a cardiologist could deliver Aortix through a catheter in the femoral artery to the descending thoracic aorta, a strategic location downstream of the heart that allows for combined benefit to the heart, kidneys, and other vital organs. Once the catheter sheath is in place, it is retracted to deploy the self-expanding nickel-titanium struts that anchor the pump to the aortic wall. |
How does your product differ from the competition? | Adhesives, tapes, and expensive securement devices have long been viewed as the last lines of defense in avoiding IV complications. While basic securement is essential, all securement devices will fail eventually, resulting in catheter dislodgment. Unlike basic securement devices, the SafeBreak IV creates a necessary separation in the IV line, allowing us to protect the IV catheter during extreme tension. The SafeBreak IV helps understaffed nurses avoid stressful and time consuming IV dislodgment, and keeps patients progressing through the continuum of care. |
| This reversible anchoring is essential to what makes Aortix the first catheter-based pump suitable for ambulatory use in a walking, active patient. Additionally, the pump's location minimizes the common VAD surgical risks and potential for thrombotic stroke. In contrast to traditional VADs, where failure is often fatal, Aortix does not obstruct native blood flow and device failure is not life threatening. Aortix is designed to augment the natural function of the heart by accelerating a portion of the body's native blood flow within the micropump and directing it downstream in a series of "jets" that entrain and accelerate the native flow. In pre-clinical animal studies, Aortix increased the amount of blood the heart pumps by 10-15% while simultaneously lowering its energy needs by almost 40%. The end result is a nearly 60% increase in efficiency, and a heart that is working at a sustainable level while providing healthy blood flow and pressure to end organs. This groundbreaking interventional cardiology tool, conceived by cardiologist Dr. Reynolds M. Delgado, III, medical director of Mechanical Support Devices in Heart Failure at the Texas Heart Institute, has the potential to replace high-risk surgical devices and lengthy hospital stays with a lower-risk outpatient cardiology procedure. This type of paradigm shift could not only improve the quality of life for millions of chronic heart failure patients, but also benefit hospitals and payers by potentially cutting treatment costs and readmission rates. |
Do you have customers yet? | The device is not yet cleared for sale by FDA, but we will start our first clinical trial in the fall of 2016. |
| Procyrion is a development-stage company, and the Aortix micro-pump is currently in preclinical testing to support a first-in-human trial scheduled for later this year (2016). Aortix is not approved for sale or use. |
How much money have you raised? | Approximately $900K |
| $13.5 million |
Who are your investors? | Zero To 510 Medical Device Accelerator, venture capital firms, and angel investors |
| Fannin Innovation Studio, Scientific Health Development, undisclosed strategic and angel investors |
What is the next milestone for your device? | FDA 510(k) clearance is the next milestone for our device. We will be submitting our 510(k) in the fall of 2016. |
| First-in-Human clinical trial scheduled for later in 2016. |
Continue to "10 Digital Health and Medtech Startups that Shone at SXSW"
You May Also Like