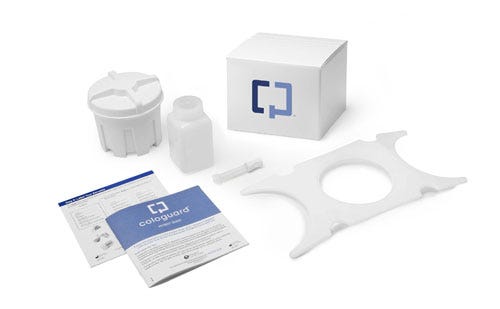
Exact Sciences In August, Exact Sciences was granted PMA approval for Cologard, the first and only FDA-approved DNA test for colorectal cancer.
September 29, 2014
Exact Sciences
|
Exact Sciences's Cologard, a DNA test for colorectal cancer, was unanimously recommended for FDA approval in March 2014. |
In August, Exact Sciences was granted PMA approval for Cologard, the first and only FDA-approved DNA test for colorectal cancer.
The test screens for both cancer and precancers—something other stool-based colorectal cancer tests on the market can’t do. But perhaps the most disruptive aspect of the test is the fact that it can be taken in the privacy of a patient’s own home. The Madison, WI-based company hopes that will encourage more people to get screened for colocrectal cancer, which is the second leading cause of cancer deaths in the United States.
Exact Sciences expects a final payment decision from CMS in October or November, at which time the product will officially launch. Mayo Clinic has already announced it will be the first healthcare system to provide Cologard for its patients, and Exact Sciences hopes the test will also be included in colorectal cancer screening guidelines issued by organizations such as the American Cancer Society by the end of 2015.
|
|
|
|
[image courtesy of EXACT SCIENCES]
You May Also Like