July 30, 2015
Minimally invasive mitral valve replacement therapies appear to be a hot area for medical device companies.
Chris Newmarker
|
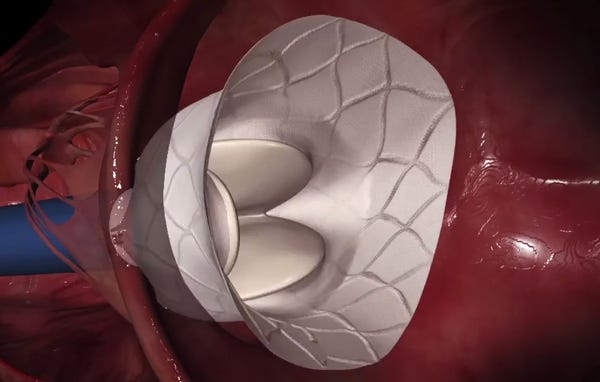
This screen grab from an animation shows the Tendyne Bioprosthetic Mitral Valve deploying. (Image courtesy of Tendyne Holdings) |
Abbott is spending $225 million to fully acquire Roseville, MN-based Tendyne Holdings and its minimally invasive mitral valve replacement technology, Abbott announced Thursday.
In a separate transaction, Abbott is spending an undisclosed sum to secure an option to buy Cephea Valve Technologies (San Jose, CA), which has been developing a catheter-based mitral valve replacement therapy.
For its part, Abbott already has its MitraClip, a mitral valve repair device used to treat certain types of mitral valve disease. It is delivered to the heart via a catheter through the femoral vein. MitraClip is available in the United States, Canada, Europe, and countries in Asia and Latin America, with more than 25,000 people treated.
"The Tendyne acquisition and our agreement with Cephea broaden our foundation as one of the leaders in treatments for mitral valve disease, with the goal of bringing promising, less invasive valve treatment technologies to people who need them," John M. Capek, PhD, executive vice president of ventures at Abbott, said in a news release.
The news comes more than two weeks after Edwards Lifesciences said it would spend up to $400 million to acquire Irvine, CA-based CardiAQ Valve Technologies and its transcatheter mitral valve replacement system.
Both Edwards and Dublin, Ireland-based Medtronic already sell minimally invasive aortic valve replacements in the U.S., with the aortic valve replacements used on patients considered too high risk for traditional open-heart surgery.
The new Abbott and Edwards deals indicate that minimally invasive mitral valve replacements are seen as an attractive area for catheter-based heart valve replacement technologies. Abbott notes that mitral valve disease is the most prevalent heart valve disease, with minimally invasive mitral valve repair and replacement expected to become a multi-billion dollar market over the next 10 years.
The Tendyne Bioprosthetic Mitral Valve System is an investigational device that already has FDA approval for a feasibility clinical trial. The trial has begun enrolling patients, with plans to start enrollment next year for a clinical trial to support CE Mark in Europe.
Add in what Abbott has already invested in Tendyne, and the total value of the deal is $250 million, plus potential future payments tied to regulatory milestones.
Refresh your medical device industry knowledge at MEDevice San Diego, September 1-2, 2015. |
Chris Newmarker is senior editor of Qmed and MPMN. Follow him on Twitter at @newmarker.
Like what you're reading? Subscribe to our daily e-newsletter.
About the Author(s)
You May Also Like